General Information
Group Leader
Prof. Dr. med. Tobias Cantz
e-mail: Cantz.Tobias(at)mh-hannover.de
Phone: +49 511 532-5251
Institution
Medizinische Hochschule Hannover and Max Planck Institute for Molecular Biomedicine, Münster
Department
Department of Gastroenterology, Hepatology and Endocrinology and MPI-Cell and Developmental Biology
Head of Department
Prof. Dr. Michael Manns (MHH) and Prof. Dr. Hans R. Schöler (MPI)
Location of Workgroup
Medizinische Hochschule Hannover
Hans-Borst-Zentrum (J11)
Carl-Neuberg-Str. 1
30625 Hannover

Further Information
- Founding member of the German Stem Cell Network and leader of the work group “outreach activities” and "Ethical, legel & social aspects": www.gscn.org
- Contribution to the public stem cell debate: Interdisciplinary project for high school teachers: www.zellux.net
- Max Planck Institute for molecular Biomedicine: www.mpi-muenster.mpg.de
Staff
| Cantz, Tobias Prof. Dr. | Cantz.Tobias(at)mh-hannover.de |
| Eggenschwiler, Reto Dr. | Eggenschwiler.Reto(at)mh-hannover.de |
| Sgodda, Malte Dr. | Sgodda.Malte(at)mh-hannover.de |
| Ebert, Katharina | Ebert.Katharina(at)mh-hannover.de |
| Papp, Oliver | Papp.Oliver(at)mh-hannover.de |
| Schneider, Malte | Schneider.Malte(at)mh-hannover.de |
| Sgodda, Susan | Sgodda.Susan(at)mh-hannover.de |
| Jahn, Christopher | Jahn.Christopher(at)mh-hannover.de |
| Alfken, Susanne | Alfken.Susanne(at)mh-hannover.de |
| Cortnumme Leon | Cortnumme.Leon(at)mh-hannover.de |
| Helli, Mahmoud | Helli.Mahmoud(at)mh-hannover.de |
| Schwarzer, Helmut | Schwarzer.Helmut(at)mh-hannover.de |
| Winter, Iris | Winter.I(at)mh-hannover.de |
Translational Hepatology and Stem Cell Biology
Objectives
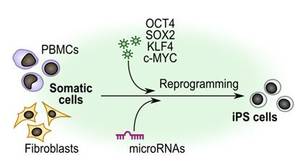
The primary focus of our group is to develop and apply improved protocols for the generation of human induced pluripotent stem cells (iPS) to provide patient-specific stem cells that can be investigated as surrogate for the diseased cell phenotype. Interested in the pathophysiology of liver disease, we are also refining differentiation protocols, which will provide hepatocyte- and cholangiocyte-like cells for studies of liver regeneration and on the long term perspective for stem cell-based therapies. As comprehensive approach, we are evaluating direct reprogramming strategies for the generation of hepatic cells for disease modeling approaches and for putative therapeutic applications.
The Research Group (RG) Translational Hepatology and Stem Cell Biology is embedded in the Department of Gastroenterology, Hepatology, and Endocrinology and is also part of the Max Planck Institute for Molecular Biomedicine in Münster contributing to the Excellence Cluster “REBIRTH” (Regenerative Biology and Reconstructive Therapy) of Hannover Medical School (MHH) funded by the DFG since October 2006
Research Focus
Molecular insights into defined iPS cell generation
The generation of patient-derived induced pluripotent stem cells (iPS cells) has opened new avenues in regenerative medicine, but some recent publications have challenged the clinical utility of iPS cells because of genomic and epigenetic abnormalities. In light of this issue, we evaluate the safe and efficient generation of iPS cells from limited sample materials (such as cryopreserved blood samples) using a polycistronic lentiviral reprogramming vector for the expression of Oct4, Sox2, KLf4, and the miR290 cluster. Such a system can avoid the expression of the known oncogene c-Myc and should nevertheless be sufficiently efficient for the generation of iPS cells from limited clinical specimens. At the molecular level, we were able to elucidate the role of other microRNAs in the generation of murine iPSCs with a focus on the miR212 / 130 family and the miR29 family (Fraguas, 2016; Pfaff, 2017).

Precise genome editing of iPS cells
The CRIPSR/Cas9 technology supports simple and accurate HDR-mediated DNA modification, and we have established a complex selection/counterselection system for efficient bi-allelic modification of the transcriptionally silent SERPINA1 locus in human iPS cells derived from a patient with severe α1-antitrypsin deficiency (AATD). For the selection of the most suitable guideRNAs, we have established a highly sensitive reporter assay that allows an in-cellam assay to evaluate the HDR efficiencies of various guideRNA combinations at their on- and off-target loci. As a proof-of-concept, this guideRNA / Cas9-nickase combination allowed us to easily identify biallelic repaired iPS cell clones that exhibit intact α1-antitrypsin expression and function (Eggenschwiler, 2016).

Hepatic cells derived from human ES- and iPS-cells
Current standard protocols for differentiation of human iPS cells into liver cells produce rather heterogeneous cell populations that require further purification, such as selection with antibiotic resistance genes (Sgodda, 2013). In further work, by modulating miR199a expression, we achieved a more mature hepatic phenotype, resulting in improved post-transplantation repopulation efficiency in FAH-/- Rag2-/- gamma-c-/- mice (Möbus, 2015). In addition, we investigated the modulation of the Wnt signaling pathway by activation by the GSK3β inhibitor CHIR99021 and subsequent inhibition by sFRP5 during the endoderm specification of human pluripotent stem cells to obtain a more homogeneous cell population with definitive endoderm properties for subsequent differentiation steps (Hoepfner, 2016). Together with Amar Deep Sharma we work on the ‘Elucidation of supporting microRNAs during the hepatic specification of reprogrammed cells in liver cell therapy’ in the colaborative research centre (SFB) 738 ‘Optimization of conventional and innovative transplants’ Another important aspect is the establishment of three-dimensional cell culture conditions, which allow a spatial aggregation of cells and thus a more authentic tissue-like organization of the differentiated cells. In close cooperation with Robert Zweigerdt (LEBAO, Department of Cardiothoracic, Transplantation and Vascular Surgery, MHH), we were able to evaluate a scalable 3D suspension culture system (500μl to 15ml) containing hepatically differentiated human pluripotent stem cells exhibiting stable liver metabolism and sensitive liver toxicity properties for up to three weeks (Sgodda, 2017). Furthermore, direct reprogramming of somatic cells is a fascinating opportunity to generate suitable cell sources for tissue and cell replacement therapy. We have contributed to a study by Dong Wook Han (Konkuk University, Seoul, South Korea) of the (short-term) in vivo potential of in vitro generated iHeps in FAH-/- Rag2-/- c-/- mice (Kim, 2015; Lim, 2016). In close collaboration with Amar Deep Sharma and Michael Ott, we were able to demonstrate in vivo reprogramming of hepatic myofibroblasts into functional hepatocytes. For this work, we used two different mouse models for lineage tracing to demonstrate the generation of iHeps from activated myofibroblasts that expressed the transcription factors FOXA3, GATA4, HNF1A, and HNF4A relevant to transcription through a cell-type specific adenoviral vector (Song, 2016).

iPSC-based disease modeling
In diseases that show broad patient-specific variability in terms of penetrance, time and organ specificity, iPS cell-based models represent a new resource for testing innovative therapeutic strategies in vitro. In a collaboration project with Hartmut Schmidt (Clinic for Transplantation Medicine, WWU Münster), we were able to produce iPS-hepatocytes (iPS-Hep) from patients with different mutations in the transthyretin gene and to establish a high-throughput screening-capable in-vitro platform for the evaluation of drugs. We aimed to model transthyretin-mediated familial amyloid polyneuropathy (TTR-FAP) with patient-specific iPS cells and differentiated them into hepatocyte-like cells. As expected, the TTR-FAP-derived liver cells showed no altered functional hepatic properties (TTR and albumin secretion as well as cytochrome P450 activity), and by mass spectrometry the mutant TTR protein (valine 30 methionine or glycine 47 alanine substitution) could be detected. The resulting amyloid fibril formation could be detected specifically by staining with intercalating dyes such as Congo Red and Thioflavin T and was also detectable in smaller cell culture formats (up to 96-well format), which would allow future use in drug screening systems (Hoepfner, 2017 ).

www.zellux.net
Can we sacrifice embryonic human life for a good purpose? This and many other questions arise when we think deeply about stem cell research. The internet portal at www.zellux.net elucidates this complex topic and provides a variety of materials for school teachers. Interested laypersons will also find a wealth of valuable information. The varying views of the religions, different philosophic traditions, and diverse political opinions are illustrated in a clear format and the portal provides resource material to form one's own ethical judgments.
In the funding priority "Ethical and Legal Aspects of the Life Sciences of the BMBF we are currently working on a project on “Human Artificial Gametes” (HumArGam) and on the precise genome editing of hereditary diseases (REALiGN-HD), in which we discuss relevant aspects of the generation of artificial germ cells and genetic engineerung in an interdisciplinary consortium with bioethicists (Thomas Heinemann, PTH Vallendar) and lawyers (Hans-Georg Dederer, University of Passau) in order to further develop an appropriate normative framework.
Collaborations
- A. Schambach, Hannover Medical School, REBIRTH Unit Regenerative Gene Therapy: Improved viral systems for the generation of iPS cells
- M. Ott, Hannover Medical School, REBIRTH Unit Hepatic Cell Transplantation and Genetic Manipulation: in vivo evaluation of pluripotent cell-derived hepatic cells
- U. Martin, Hannover Medical School, LEBAO, REBIRTH Unit iPSCs for Disease Modelling, Drug Screening and Cell Therapy: Cellular Approaches for Rare Pulmonary Diseases (CARPuD)
- T. Moritz, REBIRTH Unit iPSCs based Haematopoietic Regeneration: Generation of blood cell- and bone marrow-derived iPS cells
- Hartmut Schmidt, University Hospital Münster, Department for Transplantation Medicine: iPS-based modeling of TTR amyloidosis using urine-derived iPSCs
- Dong Wook Han, Konkuk University Seoul, South Korea: Direct transprogramming of somatic cells into hepatic cells.
- Guangming Wu, Max Planck Institute for Molecular Biomedicine, Münster: Murine embryo manipulation technologies for studying gene correction strategies in iPSCs
- Reinhard Henschler, Transfusion Service, Suisse Cross Red (SCR) Zurich: iPSC-derived mesenchymal stroma cells as immunmodulatory cell transplant
- T. Heinemann, PTH Vallendar, Dept. of Medical Ethics: bioethics
- H.-G. Dederer, University of Passau, Dept. of Law: bioethics
Further Research Projects
- SFB738 / CRC738 (Project C12): Elucidation of supportive microRNAs during hepatic specification of reprogrammed cells in liver cell therapy
- R2N: „Replace“ and „Reduce“ based in Lower Saxony: Alternative methods to replace or reduce animal models in biomedical research; Bproject B2: ‘human iPSC-derived functional hepatic cells as alternative for animal experiments in toxicology studies’
- HumArGam: Humane Artifizielle Gameten. Erzeugung und genetische Veränderung von aus humanen pluripotenten Stammzellen differenzierten Gameten und ihre ethische und rechtliche Bewertung
- REALiGN-HD: Ethische und rechtliche Konzepte für die Anwendung neuer Techniken einer präzisen Genomeditierung bei hereditären Erkrankungen
Teaching
- Lecturer in the Ph.D. programme ‘Regenerative Sciences’
- Co-supervisor of various Ph.D. students
- Consultant at school projects addressing stem cell research (Cooperation with the evang. Kirche von Westfalen)
Publications
2013 - ongoing
2018
Beh-Pajooh A, Cantz T (2018). The Role of MicroRNAs in Embryonic and Induced Pluripotency. J Stem Cells Regen Med. accepted
Advena-Regnery B, Dederer HG, Enghofer F, Cantz T, Heinemann T. Framing the ethical and legal issues of human artificial gametes in research, therapy, and assisted reproduction: A German perspective (2018). Bioethics in press
Galonska C, Charlton J, Mattei AL, Donaghey J, Clement D, Gu H, MohammadA, Stamenova E, Cacchiarelli, Klages S, Timmermann B, Cantz T, Schöler HR, Gnirke A, Ziller MJ, Meissner A. Genome-wide tracking of dCas9-methyltransferase footprints. Nat Commun 9(1):597.
2017
Pfaff N, Liebhaber S, Mobus S, Beh-Pajooh A, Fiedler J, Pfanne A, Schambach A, Thum T, Cantz T, Moritz T. Inhibition of miRNA-212/132 improves the reprogramming of fibroblasts into induced pluripotent stem cells by de-repressing important epigenetic remodelling factors. Stem Cell Res. 20:70-5.
Pittermann E, Lachmann N, MacLean G, Emmrich S, Ackermann M, Göhring G, Schlegelberger B, Welte K, Schambach A, Heckl D, Orkin SH, Cantz T, Klusmann J-H. Gene Correction of Hax1 Reversed Kostmann Disease Phenotype in Patient-Specific Induced Pluripotent Stem Cells. Blood Advances. 2017;903-14.
Sgodda M, Dai Z, Zweigerdt R, Sharma AD, Ott M, Cantz T. A Scalable Approach for the Generation of Human Pluripotent Stem Cell-Derived Hepatic Organoids with Sensitive Hepatotoxicity Features. Stem Cells Dev. 2017/07/13 ed2017. p. 1490-504.
Moslem M, Eggenschwiler R, Wichmann C, Buhmann R, Cantz T, Henschler R. Kindlin-2 Modulates the Survival, Differentiation, and Migration of Induced Pluripotent Cell-Derived Mesenchymal Stromal Cells. Stem Cells Int. 2017;2017:7316354.
Hoepfner J, Kleinsorge M, Papp O, Alfken S, Heiringhoff R, Pich A, Sauer V, Zibert A, Gohring G, Schmidt H, Sgodda M, Cantz T. In vitro modelling of familial amyloidotic polyneuropathy allows quantitative detection of transthyretin amyloid fibril-like structures in hepatic derivatives of patient-specific induced pluripotent stem cells. Biol Chem. 2017.
2016
Yang D, Yuan Q, Balakrishnan A, Bantel H, Klusmann JH, Manns MP, Ott M, Cantz T, Sharma AD. MicroRNA-125b-5p mimic inhibits acute liver failure. Nature communications. 2016;7:11916.
Thiesler CT, Cajic S, Hoffmann D, Thiel C, van Diepen L, Hennig R, Sgodda M, Weibetamann R, Reichl U, Steinemann D, Diekmann U, Huber NM, Oberbeck A, Cantz T, Kuss AW, Korner C, Schambach A, Rapp E, Buettner FF. Glycomic Characterization of Induced Pluripotent Stem Cells Derived from a Patient Suffering from Phosphomannomutase 2 Congenital Disorder of Glycosylation (PMM2-CDG). Molecular & cellular proteomics : MCP. 2016;15(4):1435-52.
Song G, Pacher M, Balakrishnan A, Yuan Q, Tsay HC, Yang D, Reetz J, Brandes S, Dai Z, Putzer BM, Arauzo-Bravo MJ, Steinemann D, Luedde T, Schwabe RF, Manns MP, Scholer HR, Schambach A, Cantz T, Ott M, Sharma AD. Direct Reprogramming of Hepatic Myofibroblasts into Hepatocytes In Vivo Attenuates Liver Fibrosis. Cell stem cell. 2016;18(6):797-808.
Lim KT, Lee SC, Gao Y, Kim KP, Song G, An SY, Adachi K, Jang YJ, Kim J, Oh KJ, Kwak TH, Hwang SI, You JS, Ko K, Koo SH, Sharma AD, Kim JH, Hui L, Cantz T, Scholer HR, Han DW. Small Molecules Facilitate Single Factor-Mediated Hepatic Reprogramming. Cell reports. 2016.
Hoepfner J, Kleinsorge M, Papp O, Ackermann M, Alfken S, Rinas U, Solodenko W, Kirschning A, Sgodda M, Cantz T. Biphasic modulation of Wnt signaling supports efficient foregut endoderm formation from human pluripotent stem cells. Cell Biol Int. 2016;40(5):534-48.
Fraguas MS, Eggenschwiler R, Hoepfner J, Schiavinato JL, Haddad R, Oliveira LH, Araujo AG, Zago MA, Panepucci RA, Cantz T. MicroRNA-29 impairs the early phase of reprogramming process by targeting active DNA demethylation enzymes and Wnt signaling. Stem cell research. 2016;19:21-30.
Eggenschwiler R, Moslem M, Fraguas MS, Galla M, Papp O, Naujock M, Fonfara I, Gensch I, Wahner A, Beh-Pajooh A, Mussolino C, Tauscher M, Steinemann D, Wegner F, Petri S, Schambach A, Charpentier E, Cathomen T, Cantz T. Improved bi-allelic modification of a transcriptionally silent locus in patient-derived iPSC by Cas9 nickase. Scientific reports. 2016;6:38198.
2015
Advena-Regnery B, Böhm K, Jung B, Rottländer K, Sgodda S. Sind Parthenoten Embryonen? Zur biologischen und normativen Einordnung von menschlichen Parthenoten. Zeitschrift für medizinische Ethik.2015. 61:63-79.
Kim J, Kim KP, Lim KT, Lee SC, Yoon J, Song G, Hwang SI, Scholer HR, Cantz T, Han DW. Generation of Integration-Free Induced Hepatocyte-Like Cells from Mouse Fibroblasts. Sci Rep. 2015;5:15706.
Katsirntaki K, Mauritz C, Olmer R, Schmeckebier S, Sgodda M, Puppe V, Eggenschwiler R, Duerr J, Schubert SC, Schmiedl A, Ochs M, Cantz T, Salwig I, Szibor M, Braun T, Rathert C, Martens A, Mall MA, Martin U. Bronchoalveolar Sublineage Specification of Pluripotent Stem Cells: Effect of Dexamethasone Plus Camp-Elevating Agents and Keratinocyte Growth Factor. Tissue Eng Part A. 2015;21(3-4):669-82.
Mobus S, Yang D, Yuan Q, Ludtke TH, Balakrishnan A, Sgodda M, Rani B, Kispert A, Arauzo-Bravo MJ, Vogel A, Manns MP, Ott M, Cantz T, Sharma AD. Microrna-199a-5p Inhibition Enhances the Liver Repopulation Ability of Human Embryonic Stem Cell-Derived Hepatic Cells. J Hepatol. 2015;62(1):101-10.
Cantz T, Sharma AD, Ott M. Concise Review: Cell Therapies for Hereditary Metabolic Liver Diseases-Concepts, Clinical Results, and Future Developments. Stem Cells. 2015;33(4):1055-62.
Moslem M, Eberle I, Weber I, Henschler R, Cantz T. Mesenchymal Stem/Stromal Cells Derived from Induced Pluripotent Stem Cells Support Cd34(Pos) Hematopoietic Stem Cell Propagation and Suppress Inflammatory Reaction. Stem Cells Int. 2015;2015:843058. Epub 2015/07/18.
Rahman SH, Kuehle J, Reimann C, Mlambo T, Alzubi J, Maeder ML, Riedel H, Fisch P, Cantz T, Rudolph C, Mussolino C, Joung JK, Schambach A, Cathomen T. Rescue of DNA-Pk Signaling and T-Cell Differentiation by Targeted Genome Editing in a Prkdc Deficient Ipsc Disease Model. PLoS Genet. 2015;11(5):e1005239.
2014
Cantz T. Die Erzeugung von Gameten aus pluripotenten Stammzellen. In: Heinemann T, Dederer H-G, Cantz T, editors. Entwicklungsbiologische Totipotenz in Ethik und Recht. Göttingen: V&R unipress; 2014. p. 57-65.
Sgodda S. Das Kriterium der Pluripotenz aus naturwissenschaftlicher Perspektive. In: Heinemann T, Dederer HG, editors. Entwicklungsbiologische Totipotenz in Ethik und Recht. Göttingen: V&R unipress; 2014. p. 13-55.
Ott M, Cantz T, Schneider A, Manns MP. [Diagnosis and Therapies for Acute Liver Failure: Scientific Developments]. Internist (Berl). 2014;55(11):1288-95. Diagnostik und Behandlung des akuten Leberversagens: Wissenschaftliche Entwicklungen.
Lachmann N, Happle C, Ackermann M, Luttge D, Wetzke M, Merkert S, Hetzel M, Kensah G, Jara-Avaca M, Mucci A, Skuljec J, Dittrich AM, Pfaff N, Brennig S, Schambach A, Steinemann D, Gohring G, Cantz T, Martin U, Schwerk N, Hansen G, Moritz T. Gene Correction of Human Induced Pluripotent Stem Cells Repairs the Cellular Phenotype in Pulmonary Alveolar Proteinosis. Am J Respir Crit Care Med. 2014;189(2):167-82.
Kuehle J, Turan S, Cantz T, Hoffmann D, Suerth JD, Maetzig T, Zychlinski D, Klein C, Steinemann D, Baum C, Bode J, Schambach A. Modified Lentiviral Ltrs Allow Flp Recombinase-Mediated Cassette Exchange and in Vivo Tracing of "Factor-Free" Induced Pluripotent Stem Cells. Mol Ther. 2014;22(5):919-28.
Schott JW, Hoffmann D, Maetzig T, Muller FJ, Steinemann D, Zychlinski D, Cantz T, Baum C, Schambach A. Improved Retroviral Episome Transfer of Transcription Factors Enables Sustained Cell Fate Modification. Gene Ther. 2014;21(11):938-49.
Ackermann M, Lachmann N, Hartung S, Eggenschwiler R, Pfaff N, Happle C, Mucci A, Gohring G, Niemann H, Hansen G, Schambach A, Cantz T, Zweigerdt R, Moritz T. Promoter and Lineage Independent Anti-Silencing Activity of the A2 Ubiquitous Chromatin Opening Element for Optimized Human Pluripotent Stem Cell-Based Gene Therapy. Biomaterials. 2014;35(5):1531-42.
2013
Eberle I, Moslem M, Henschler R, Cantz T. Engineered Mscs from Patient-Specific Ips Cells. Adv Biochem Eng Biotechnol. 2013;130:1-17.
Pfaff N, Cantz T. From Skin to Blood: A New Member Joins the Iclub. Cell Stem Cell. 2013;13(2):131-3.
Yuan Q, Loya K, Rani B, Mobus S, Balakrishnan A, Lamle J, Cathomen T, Vogel A, Manns MP, Ott M, Cantz T, Sharma AD. Microrna-221 Overexpression Accelerates Hepatocyte Proliferation During Liver Regeneration. Hepatology. 2013;57(1):299-310.
Gross B, Sgodda M, Rasche M, Schambach A, Gohring G, Schlegelberger B, Greber B, Linden T, Reinhardt D, Cantz T, Klusmann JH. Improved Generation of Patient-Specific Induced Pluripotent Stem Cells Using a Chemically-Defined and Matrigel-Based Approach. Curr Mol Med. 2013;13(5):765-76. Epub 2013/05/07.
Sgodda M, Mobus S, Hoepfner J, Sharma AD, Schambach A, Greber B, Ott M, Cantz T. Improved Hepatic Differentiation Strategies for Human Induced Pluripotent Stem Cells. Curr Mol Med. 2013;13(5):842-55.
Schmeckebier S, Mauritz C, Katsirntaki K, Sgodda M, Puppe V, Duerr J, Schubert SC, Schmiedl A, Lin Q, Palecek J, Draeger G, Ochs M, Zenke M, Cantz T, Mall MA, Martin U. Keratinocyte Growth Factor and Dexamethasone Plus Elevated Camp Levels Synergistically Support Pluripotent Stem Cell Differentiation into Alveolar Epithelial Type Ii Cells. Tissue Eng Part A. 2013;19(7-8):938-51.
Grabundzija I, Wang J, Sebe A, Erdei Z, Kajdi R, Devaraj A, Steinemann D, Szuhai K, Stein U, Cantz T, Schambach A, Baum C, Izsvak Z, Sarkadi B, Ivics Z. Sleeping Beauty Transposon-Based System for Cellular Reprogramming and Targeted Gene Insertion in Induced Pluripotent Stem Cells. Nucleic Acids Res. 2013;41(3):1829-47.
Sgodda M, Cantz T. Small but Significant: Inter- and Intrapatient Variations in Ips Cell-Based Disease Modeling. Mol Ther. 2013;21(1):5-7.
Eggenschwiler R, Loya K, Wu G, Sharma AD, Sgodda M, Zychlinski D, Herr C, Steinemann D, Teckman J, Bals R, Ott M, Schambach A, Scholer HR, Cantz T. Sustained Knockdown of a Disease-Causing Gene in Patient-Specific Induced Pluripotent Stem Cells Using Lentiviral Vector-Based Gene Therapy. Stem Cells Transl Med. 2013;2(9):641-54.
Pfaff N, Lachmann N, Ackermann M, Kohlscheen S, Brendel C, Maetzig T, Niemann H, Antoniou MN, Grez M, Schambach A, Cantz T, Moritz T. A Ubiquitous Chromatin Opening Element Prevents Transgene Silencing in Pluripotent Stem Cells and Their Differentiated Progeny. Stem Cells. 2013;31(3):488-99.
2006 - 2012
2012
Advena-Regnerv B, Laimböck L, Rottländer K, Sgodda S. Totipotenz im Spannungsfeld von Biologie, Ethik und Recht. Zeitschrift für medizinische Ethik. 2012;58(3):217-36.
Kleger A, Mahaddalkar PU, Katz SF, Lechel A, Joo JY, Loya K, Lin Q, Hartmann D, Liebau S, Kraus JM, Cantz T, Kestler HA, Zaehres H, Scholer H, Rudolph KL. Increased Reprogramming Capacity of Mouse Liver Progenitor Cells, Compared with Differentiated Liver Cells, Requires the Baf Complex. Gastroenterology. 2012;142(4):907-17.
Pfaff N, Lachmann N, Kohlscheen S, Sgodda M, Arauzo-Bravo MJ, Greber B, Kues W, Glage S, Baum C, Niemann H, Schambach A, Cantz T, Moritz T. Efficient Hematopoietic Redifferentiation of Induced Pluripotent Stem Cells Derived from Primitive Murine Bone Marrow Cells. Stem Cells Dev. 2012;21(5):689-701.
Lachmann N, Jagielska J, Heckl D, Brennig S, Pfaff N, Maetzig T, Modlich U, Cantz T, Gentner B, Schambach A, Moritz T. Microrna-150-Regulated Vectors Allow Lymphocyte-Sparing Transgene Expression in Hematopoietic Gene Therapy. Gene Ther. 2012;19(9):915-24.
Pfaff N, Moritz T, Thum T, Cantz T. Mirnas Involved in the Generation, Maintenance, and Differentiation of Pluripotent Cells. J Mol Med (Berl). 2012;90(7):747-52.
Gross B, Pittermann E, Reinhardt D, Cantz T, Klusmann JH. Prospects and Challenges of Reprogrammed Cells in Hematology and Oncology. Pediatr Hematol Oncol. 2012;29(6):507-28.
2011
Pfaff N, Fiedler J, Holzmann A, Schambach A, Moritz T, Cantz T, Thum T. Mirna Screening Reveals a New Mirna Family Stimulating Ips Cell Generation Via Regulation of Meox2. EMBO Rep. 2011;12(11):1153-9.
Terwel D, Loschmann YN, Schmidt HH, Scholer HR, Cantz T, Heneka MT. Neuroinflammatory and Behavioural Changes in the Atp7b Mutant Mouse Model of Wilson's Disease. J Neurochem. 2011;118(1):105-12.
Tiemann U, Sgodda M, Warlich E, Ballmaier M, Scholer HR, Schambach A, Cantz T. Optimal Reprogramming Factor Stoichiometry Increases Colony Numbers and Affects Molecular Characteristics of Murine Induced Pluripotent Stem Cells. Cytometry A. 2011;79(6):426-35.
Osiak A, Radecke F, Guhl E, Radecke S, Dannemann N, Lutge F, Glage S, Rudolph C, Cantz T, Schwarz K, Heilbronn R, Cathomen T. Selection-Independent Generation of Gene Knockout Mouse Embryonic Stem Cells Using Zinc-Finger Nucleases. PLoS One. 2011;6(12):e28911. Epub 2011/12/24.
Haake C, Bonk S, Parsiegla J, Tomala M, Loya K, Sgodda M, Cantz T, Schambach A, Kasper C, Scheper T. Comparison of the Activity and Pluripotency Maintaining Potential of Human Leukemia Inhibitory Factor (Lif) Produced in E.Coli and Cho Cells. BMC Proc. 2011;5 Suppl 8:P109.
Wu G, Gentile L, Do JT, Cantz T, Sutter J, Psathaki K, Arauzo-Bravo MJ, Ortmeier C, Scholer HR. Efficient Derivation of Pluripotent Stem Cells from Sirna-Mediated Cdx2-Deficient Mouse Embryos. Stem Cells Dev. 2011;20(3):485-93.
Wu G, Liu N, Rittelmeyer I, Sharma AD, Sgodda M, Zaehres H, Bleidissel M, Greber B, Gentile L, Han DW, Rudolph C, Steinemann D, Schambach A, Ott M, Scholer HR, Cantz T. Generation of Healthy Mice from Gene-Corrected Disease-Specific Induced Pluripotent Stem Cells. PLoS Biol. 2011;9(7):e1001099.
Eggenschwiler R, Loya K, Sgodda M, Andre F, Cantz T. Hepatic Differentiation of Murine Disease-Specific Induced Pluripotent Stem Cells Allows Disease Modelling in Vitro. Stem Cells Int. 2011;2011:924782.
Wurm M, Gross B, Sgodda M, Standker L, Muller T, Forssmann WG, Horn PA, Blasczyk R, Cantz T. Improved Lentiviral Gene Transfer into Human Embryonic Stem Cells Grown in Co-Culture with Murine Feeder and Stroma Cells. Biol Chem. 2011;392(10):887-95. Epub 2011/08/05.
Sharma AD, Narain N, Handel EM, Iken M, Singhal N, Cathomen T, Manns MP, Scholer HR, Ott M, Cantz T. Microrna-221 Regulates Fas-Induced Fulminant Liver Failure. Hepatology. 2011;53(5):1651-61.
Warlich E, Kuehle J, Cantz T, Brugman MH, Maetzig T, Galla M, Filipczyk AA, Halle S, Klump H, Scholer HR, Baum C, Schroeder T, Schambach A. Lentiviral Vector Design and Imaging Approaches to Visualize the Early Stages of Cellular Reprogramming. Mol Ther. 2011;19(4):782-9.
2010
Zaehres H, Kogler G, Arauzo-Bravo MJ, Bleidissel M, Santourlidis S, Weinhold S, Greber B, Kim JB, Buchheiser A, Liedtke S, Eilken HM, Graffmann N, Zhao X, Meyer J, Reinhardt P, Burr B, Waclawczyk S, Ortmeier C, Uhrberg M, Scholer HR, Cantz T, Wernet P. Induction of Pluripotency in Human Cord Blood Unrestricted Somatic Stem Cells. Exp Hematol. 2010;38(9):809-18, 18 e1-2.
Voelkel C, Galla M, Maetzig T, Warlich E, Kuehle J, Zychlinski D, Bode J, Cantz T, Schambach A, Baum C. Protein Transduction from Retroviral Gag Precursors. Proc Natl Acad Sci U S A. 2010;107(17):7805-10.
Tomala M, Lavrentieva A, Moretti P, Rinas U, Kasper C, Stahl F, Schambach A, Warlich E, Martin U, Cantz T, Scheper T. Preparation of Bioactive Soluble Human Leukemia Inhibitory Factor from Recombinant Escherichia Coli Using Thioredoxin as Fusion Partner. Protein Expr Purif. 2010;73(1):51-7.
Schambach A, Cantz T, Baum C, Cathomen T. Generation and Genetic Modification of Induced Pluripotent Stem Cells. Expert Opin Biol Ther. 2010;10(7):1089-103.
Cantz T, Martin U. Induced Pluripotent Stem Cells: Characteristics and Perspectives. Adv Biochem Eng Biotechnol. 2010;123:107-26.
2009
Kriegeskotte C, Cantz T, Haberland J, Zibert A, Haier J, Kohler G, Scholer HR, Schmidt HH, Arlinghaus HF. Laser Secondary Neutral Mass Spectrometry for Copper Detection in Micro-Scale Biopsies. J Mass Spectrom. 2009;44(10):1417-22.
Haridass D, Yuan Q, Becker PD, Cantz T, Iken M, Rothe M, Narain N, Bock M, Norder M, Legrand N, Wedemeyer H, Weijer K, Spits H, Manns MP, Cai J, Deng H, Di Santo JP, Guzman CA, Ott M. Repopulation Efficiencies of Adult Hepatocytes, Fetal Liver Progenitor Cells, and Embryonic Stem Cell-Derived Hepatic Cells in Albumin-Promoter-Enhancer Urokinase-Type Plasminogen Activator Mice. Am J Pathol. 2009;175(4):1483-92.
Loya K, Eggenschwiler R, Ko K, Sgodda M, Andre F, Bleidissel M, Scholer HR, Cantz T. Hepatic Differentiation of Pluripotent Stem Cells. Biol Chem. 2009;390(10):1047-55.
2008
Sancho-Bru P, Najimi M, Caruso M, Pauwelyn K, Cantz T, Forbes S, Roskams T, Ott M, Gehling U, Sokal E, Verfaillie CM, Muraca M. Stem and Progenitor Cells for Liver Repopulation: Can We Standardise the Process from Bench to Bedside? Gut. 2009;58(4):594-603. Epub 2008/12/19.
Cantz T, Key G, Bleidissel M, Gentile L, Han DW, Brenne A, Scholer HR. Absence of Oct4 Expression in Somatic Tumor Cell Lines. Stem Cells. 2008;26(3):692-7.
Cantz T, Bleidissel M, Stehling M, Scholer HR. In Vitro Differentiation of Reprogrammed Murine Somatic Cells into Hepatic Precursor Cells. Biol Chem. 2008;389(7):889-96. Epub 2008/07/17.
Sharma AD, Cantz T, Vogel A, Schambach A, Haridass D, Iken M, Bleidissel M, Manns MP, Scholer HR, Ott M. Murine Embryonic Stem Cell-Derived Hepatic Progenitor Cells Engraft in Recipient Livers with Limited Capacity of Liver Tissue Formation. Cell Transplant. 2008;17(3):313-23.